Since the introduction of the drug Gleevec® and similar tyrosine kinase inhibitors, the number of patients with chronic myeloid leukemia (CML) who enter remission and stay in remission for years has increased dramatically over the past decade.
For survival statistics, click here.
Treatment Adherence
It is important for patients to continue taking their medication to get the best response. Unless they are following their doctor’s instructions, stopping medication or taking less than the amount prescribed can impact how well the medication works and may result in a loss of response and an unfavorable treatment outcome.
For patients with CML, adhering to treatment is associated with the probability of achieving and improving long-term outcomes, including achieving a major molecular response and improved survival. Good adherence means that a patient follows all of the doctor’s recommendations, including
- Taking the appropriate number of pills as ordered: at the right time, on the right day, every day
- Reporting any side effects so that supportive treatment can be administered
- Avoiding foods, liquids or other medications that not allowed.
Patients should let their treatment team members know if there are any problems (including side effects) that make it difficult for them to take the medication as prescribed. Generally, these difficulties can be either managed or completely overcome.
My CML Tracker Pages is a tool to help you keep track of appointments, questions for your doctor, medications, side effects, test results and notes. You may download a copy of My CML Tracker Pages.
Measuring Treatment Response
Your doctor needs to monitor your response to drug therapy for chronic myeloid leukemia (CML) with blood and bone marrow tests. This is a critically important part of treatment for people with CML.
Your test results help your doctor decide whether to:
- Increase your drug dose to try for a better response
- Decrease or stop the drug briefly because of side effects
- Change to a different drug or drug combination to better control the CML
- Change to a different drug or drug combination to manage side effects
Your doctor measures your response using general treatment response guidelines for your first year of CML drug therapy. He or she regularly measures the number of red cells, white cells, platelets and CML cells in your blood. Your doctor compares these numbers with the results of the lab test done at the start of your treatment.
Types of Response
After patients begin treatment, their doctors will periodically order blood and bone marrow tests to determine whether they are responding to treatment. A treatment response is an improvement related to the patient’s treatment. Monitoring treatment response is one of the key strategies for managing CML. In general, the greater the response to drug therapy, the longer the disease will be controlled
There are three types of responses:
- Hematologic
- Cytogenetic
- Molecular
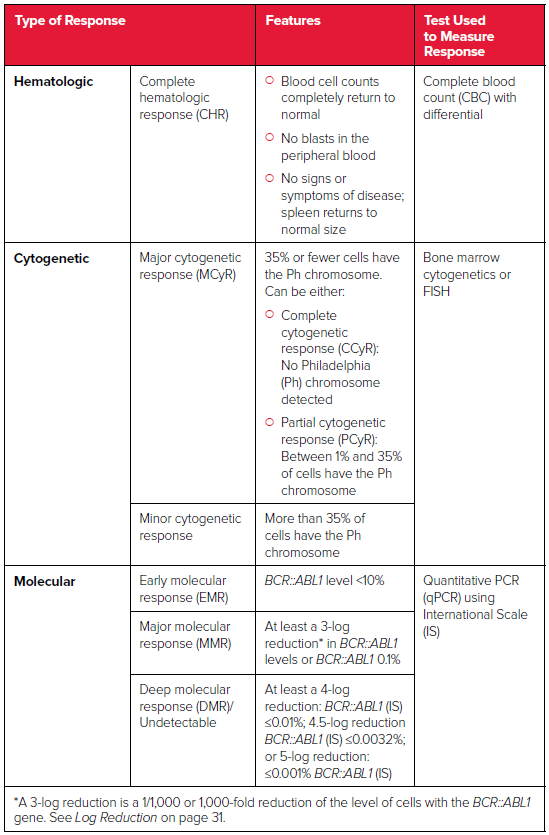
Source: National Comprehensive Cancer Network. NCCN Clinical Practice Guidelines in Oncology: Chronic Myeloid Leukemia; version 1.2023.
Treatment Response Milestones
People with CML respond to treatment in different ways. These are general guidelines for CML drug therapy. Your baseline results (your test levels at the time of diagnosis) can influence your response. But, your doctor will generally work with the following time frame as a guideline to achieve the desired response:
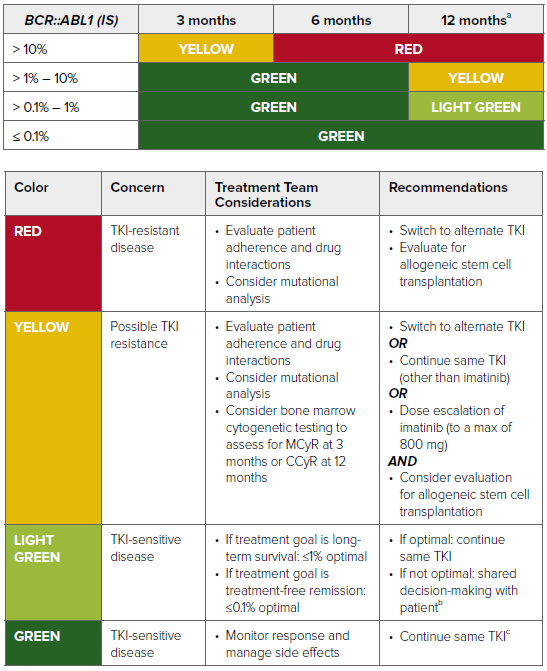
Adapted from: National Comprehensive Cancer Network. NCCN Clinical Practice Guidelines in Oncology: Chronic Myeloid Leukemia; version 1.2023.
Abbreviations: MCyR, major cytogenetic response; CCyR, complete cytogenetic response; TKI, tyrosine kinase inhibitor.
aBCR::ABL1 ≤0.1% at 12 months is associated with a very low probability of disease progression and a high likelihood of achieving a subsequent deep molecular response (MR4.0; ≤0.01% BCR::ABL1 IS), which may allow for discontinuation of TKI therapy.
bSwitching from imatinib to a second-generation TKI improves response but is associated with increased toxicity.
cDiscontinuation of TKI with careful monitoring is possible in select patients.
The International Scale (IS)
This is a standardized scale for measuring qPCR test results. The qPCR test measures the number of cells that have the BCR-ABL1 gene. It is used to determine how well treatment is working. The International Scale defines the standard baseline as BCR::ABL1 100 percent. A log reduction indicates the BCR::ABL1 level has decreased by a certain amount from the standard baseline.
- 1-log reduction means that the BCR::ABL1 level has decreased to 10 times below the standardized baseline. This means that 10 percent of cells (10 out of every 100 cells) have the BCR::ABL1 gene. This reduction is equivalent to an early molecular response when achieved within 3 to 6 months of starting treatment.
- 2-log reduction means that the BCR::ABL1 level has decreased to 100 times below the standardized baseline. This means that 1 percent of cells (1 out of every 100 cells) have the BCR::ABL1 gene.
- 3-log reduction means that the BCR::ABL1 level has decreased to 1,000 times below the standardized baseline. This means that 0.1 percent of cells (1 out of every 1,000 cells) have the BCR::ABL1 gene. It is also known as a “major molecular response (MMR).”
- 4-log reduction means that 0.01% of cells (1 out of every 10,000 cells) have the BCR::ABL1 gene.
- 4.5-log reduction is referred to as a “complete molecular response (CMR)” or a “deep molecular response (DMR).” Doctors may refer to this as “MR4.5.” A 4.5-log reduction indicates that 0.0032% of cells (1 out of every 32,000 cells) have the BCR::ABL1 gene. Achieving a deep molecular response is a sign of disease remission. Patients who achieve and then sustain a deep molecular response for a significant period of time may be considered candidates for discontinuing TKI therapy under careful medical supervision.
- 5-log reduction means that 0.001% of cells (1 out of every 100,000) have the BCR::ABL1 gene. By reaching a 5-log reduction, patients have achieved “undetectable BCR::ABL1.”
For patients, qPCR testing is recommended every 3 months initially. After 2 years of achieving and maintaining a BCR::ABL1 level of 0.1 percent of less, the test should be done every 3-6 months.
BCR::ABL1 Kinase Domain Mutation Analysis
In CML, mutations in the BCR::ABL1 gene alter the shape of the BCR::ABL1 protein, which can affect the blocking action of the TKI on BCR::ABL1, allowing cancer cells to grow again. A BCR::ABL1 kinase domain mutation analysis is a test that looks for mutations in the BCR::ABL1 gene that may cause certain TKIs to stop working. This test should be performed if there is:
- An inadequate response to the initial TKI therapy
- A failure to meet a treatment milestone
- A loss of hematologic, cytogenetic or major molecular response, or a 1-log increase in the BCR::ABL1 level, in the context of continuous therapy
- Progression to accelerated phase or blast phase CML
Mutation testing does not need to be done in patients who are switching medication because of side effects.
Among the BCR::ABL1 mutations:
- T315I is resistant to imatinib, dasatinib, nilotinib and bosutinib
- F317L and V299L are resistant to dasatinib
- Y253H, E255K/V and F359C/V are resistant to nilotinib
- T315I, G250E and V299L are resistant to bosutinib
For people with CML that stops responding to a TKI, or who do not achieve the expected response within a given period of time, the most common options are switching to another approved TKI or participating in a clinical trial..
Related Links
- Download or order The Leukemia & Lymphoma Society's free booklets:
